Pharma Manufacturing: AI for Packaging and Label Accuracy
Recent Post:
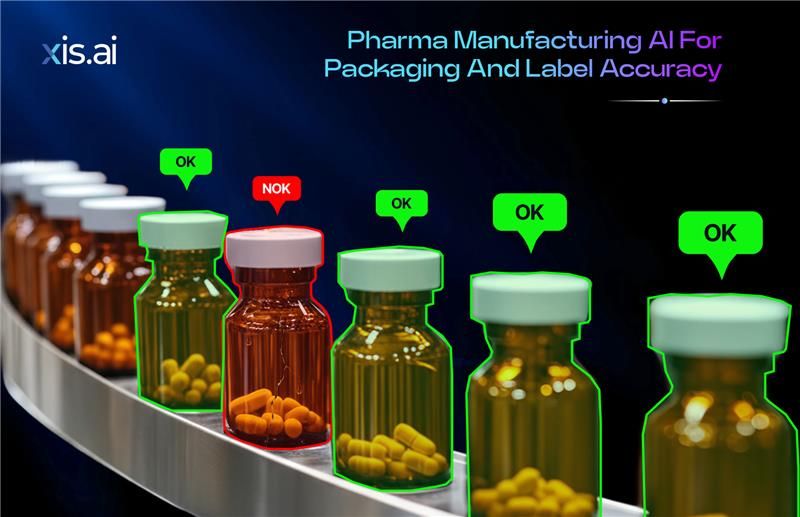
Why Packaging and Label Accuracy Is Mission-Critical in Pharma
Pharma manufacturing operates under zero-error tolerance. Every label, printed code, and packaging element must be accurate, verifiable, and compliant with serialization and traceability regulations. A single labeling mistake can lead to regulatory action, costly recalls, disrupted supply chains, and serious patient safety risks.
In modern pharmaceutical production, packaging is more than presentation—it is a regulated data carrier. Batch numbers, expiry dates, serialized QR or Data Matrix codes, dosage instructions, and tamper-evident markers all serve as compliance checkpoints. As production scales and global distribution expands, maintaining consistent inspection accuracy becomes increasingly complex.
This is where AI-powered inspection solutions from xis.ai provide measurable value.
The Growing Complexity of Pharmaceutical Packaging
Pharmaceutical packaging lines operate at high speeds while handling small, reflective, and often curved surfaces such as blister packs, vials, syringes, and cartons. Minor print defects, smudging, or misaligned labels may not be easily visible to the human eye, yet they can invalidate compliance requirements.
Traditional barcode scanners rely on ideal printing conditions. However, real-world production environments involve lighting variations, vibration, and print inconsistencies. These factors increase the risk of unreadable or mismatched codes slipping through quality checks.
xis.ai addresses this challenge by combining computer vision and deep learning to analyze packaging visually and contextually, ensuring that both the printed information and its placement meet strict pharmaceutical standards.
How xis.ai Uses AI to Improve Packaging and Label Accuracy
AI-powered inspection systems from xis.ai go beyond basic code scanning. High-resolution cameras capture detailed images of each unit in motion, and intelligent neural networks evaluate multiple parameters simultaneously.
The system verifies code readability, checks text accuracy such as batch and expiry information, detects label misalignment, and identifies subtle print defects. Even partially damaged or low-contrast codes can be interpreted accurately through advanced image reconstruction models.
More importantly, xis.ai integrates inspection with enterprise systems such as ERP and manufacturing execution platforms. This ensures that the printed serialization data matches the correct product and batch information in real time. If a mismatch is detected, automated responses can reject the unit or alert operators immediately.
This shift from passive scanning to intelligent validation significantly strengthens operational control.
Strengthening Serialization and Regulatory Compliance
Global pharmaceutical markets require strict track-and-trace compliance. Each product must carry a unique serialized identifier that remains accurate throughout its lifecycle. Any break in the traceability chain can trigger regulatory investigations or distribution delays.
xis.ai enhances serialization integrity by creating a digital audit trail for every inspected unit. Each read and validation event is logged securely, providing documentation for regulatory audits and compliance reporting. This level of traceability reduces the administrative burden while increasing confidence during inspections.
By automating verification at scale, pharmaceutical manufacturers can maintain consistent compliance across multiple production lines and facilities.
Reducing Recall Risk and Protecting Brand Integrity
Labeling errors are among the leading causes of pharmaceutical recalls. Incorrect dosage information, mismatched batch numbers, or unreadable codes can result in immediate market withdrawal. Beyond financial losses, recalls damage brand reputation and erode patient trust.
With real-time AI inspection from xis.ai, errors are detected at the production stage rather than after distribution. Intelligent systems operate continuously without fatigue, ensuring consistent oversight across high-speed packaging lines. This proactive approach reduces recall risks and supports a stronger quality assurance framework.
Enabling Smart Pharma Manufacturing
As pharmaceutical manufacturers move toward digital transformation and smart factory initiatives, AI-driven inspection becomes a strategic asset. Beyond verifying labels, AI can analyze recurring defect patterns, monitor print quality trends, and provide data-driven insights for process optimization.
xis.ai supports this evolution by delivering scalable AI solutions that integrate seamlessly into existing production environments. Whether deployed on individual packaging lines or across global facilities, the system adapts to varying formats and compliance requirements while maintaining high inspection accuracy.
This enables pharmaceutical companies to move from reactive quality control to predictive quality intelligence.
Conclusion
Pharma manufacturing demands absolute precision in packaging and labeling. As regulatory expectations intensify and production complexity increases, relying solely on traditional inspection methods introduces unnecessary risk.
xis.ai enables pharmaceutical manufacturers to implement advanced AI-powered packaging and label accuracy solutions that combine computer vision, deep learning, and real-time enterprise validation. By ensuring accurate serialization, reducing labeling errors, and strengthening audit readiness, xis.ai helps organizations protect patient safety, maintain regulatory compliance, and build resilient, future-ready production systems.
Frequently Asked Questions
How does AI improve pharmaceutical packaging accuracy?
AI uses computer vision and deep learning to verify codes, text, and label placement in real time, reducing manual inspection errors.
Can xis.ai handle high-speed packaging lines?
Yes, xis.ai systems are designed for real-time processing and operate efficiently in high-throughput pharma environments.
How does xis.ai support serialization compliance?
xis.ai validates unique product identifiers against enterprise systems and maintains a digital audit trail for regulatory documentation.
Can AI detect small print defects?
AI models can identify subtle issues such as smudging, incomplete printing, and label misalignment that traditional scanners may miss.
Is integration with existing pharma systems possible?
xis.ai solutions integrate with ERP and MES platforms to ensure synchronized data validation and compliance control.
Comment
0Comments
No comments yet.


